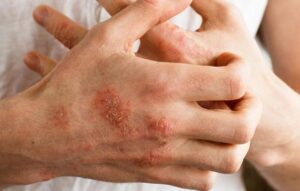
Atopic Dermatitis
RESEARCH STUDY FOR ATOPIC DERMATITIS (32 WEEKS)
Adults from 18 to less than 65 years of age having chronic atopic dermatitis (AD) with an onset of symptoms at least 3 years prior to Baseline, may qualify to receive one of two doses of investigational medication (active study drug).
To be eligible, participants must have moderate to severe AD with involvement of 10% or more body surface area (BSA). Qualified participants must be candidates for systemic treatment defined as prior use of systemic treatment for AD, or previous inadequate response to topical corticosteroids (TCS), topical calcineurin inhibitors (TCI) or PDE-4 inhibitors, or for whom topical treatments are otherwise medically inadvisable.
Eligible participants will have regular visits with an experienced research team. Qualified participants will not be charged for participating, and participants will be reimbursed for some study-related expenses. Individuals will be asked to visit the study centre about 7 times over a period of about 32 weeks.
For more information, please contact:
Principal Investigator
A/Prof Peter Foley
Sub Investigator
Dr Jackie Nguyen
t: 9623 9464
e: [email protected]
Study Coordinator
Tracy Mallett: 9623 9406
e: [email protected]

